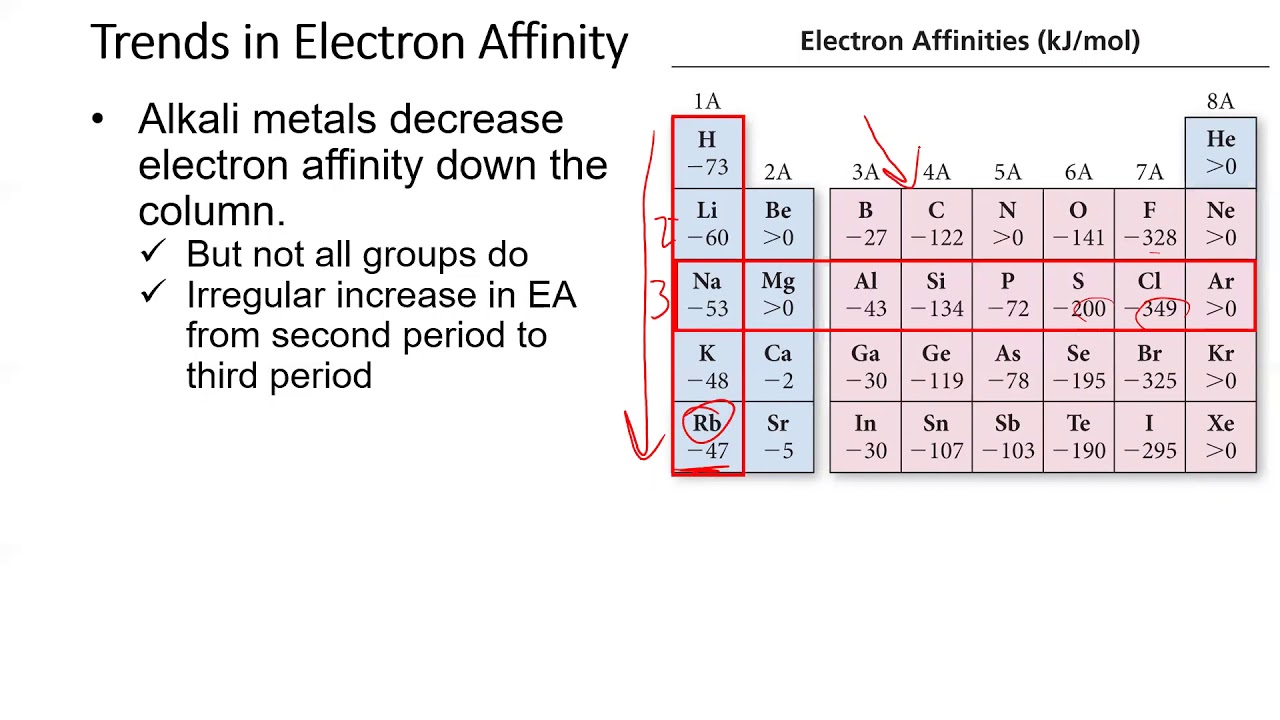
Also shown: Fermi level E F, valence band edge E V, work function W. Thus, electron affinity follows the same "left-right" trend as electronegativity, but not the "up-down" trend.īand diagram of semiconductor-vacuum interface showing electron affinity E EA, defined as the difference between near-surface vacuum energy E vac, and near-surface conduction band edge E C. For example, E ea actually increases consistently on descending the column for the group 2 data. In group 18, the valence shell is full, meaning that added electrons are unstable, tending to be ejected very quickly.Ĭounterintuitively, E ea does not decrease when progressing down most columns of the periodic table. 
This is caused by the filling of the valence shell of the atom a group 17 atom releases more energy than a group 1 atom on gaining an electron because it obtains a filled valence shell and therefore is more stable. The electron affinities of the noble gases have not been conclusively measured, so they may or may not have slightly negative values.Į ea generally increases across a period (row) in the periodic table prior to reaching group 18. 
Chlorine most strongly attracts extra electrons neon most weakly attracts an extra electron. Atoms whose anions are more stable than neutral atoms have a greater E ea. Generally, nonmetals have more positive E ea than metals. Main article: Electron affinity (data page)Īlthough E ea varies greatly across the periodic table, some patterns emerge. Since almost all detachments (require +) an amount of energy listed on the table, those detachment reactions are endothermic, or Δ E(detach) > 0.Į ea = ( E final − E initial) detach = Δ E(detach) = −Δ E(attach). If the same table is employed for the forward and reverse reactions, without switching signs, care must be taken to apply the correct definition to the corresponding direction, attachment (release) or detachment (require). This expression does follow the convention Δ X = X(final) − X(initial) since −Δ E = −( E(final) − E(initial)) = E(initial) − E(final).Įquivalently, electron affinity can also be defined as the amount of energy required to detach an electron from the atom while it holds a single-excess-electron thus making the atom a negative ion, i.e. The usual expression for calculating E ea when an electron is attached isĮ ea = ( E initial − E final) attach = −Δ E(attach) Negative values typically arise for the capture of a second electron, but also for the nitrogen atom. In this case, the electron capture is an endothermic process and the relationship, E ea = −Δ E(attach) is still valid. However, if the value assigned to E ea is negative, the negative sign implies a reversal of direction, and energy is required to attach an electron. The relation between the two is E ea = −Δ E(attach). Confusion arises in mistaking E ea for a change in energy, Δ E, in which case the positive values listed in tables would be for an endo- not exo-thermic process. It is the word "released" within the definition "energy released" that supplies the negative sign to Δ E. The positive values that are listed in tables of E ea are amounts or magnitudes. Electron capture for almost all non- noble gas atoms involves the release of energy and thus are exothermic. For any reaction that releases energy, the change Δ E in total energy has a negative value and the reaction is called an exothermic process. To use electron affinities properly, it is essential to keep track of sign. Together they may undergo charge-transfer reactions. Another example, a molecule or atom that has a more positive value of electron affinity than another is often called an electron acceptor and the less positive an electron donor. Other theoretical concepts that use electron affinity include electronic chemical potential and chemical hardness. Mulliken to develop an electronegativity scale for atoms, equal to the average of the electronsĪffinity and ionization potential. This property is used to measure atoms and molecules in the gaseous state only, since in a solid or liquid state their energy levels would be changed by contact with other atoms or molecules.Ī list of the electron affinities was used by Robert S. Measurement and use of electron affinity 4 "Electron affinity" as defined in solid state physics.1 Measurement and use of electron affinity.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
